Article: Budding Cells
Budding Yeast Cells – Why They Matter and Why You Should Count Them
by Stephen Staples and Dr. Ulrich Tillich
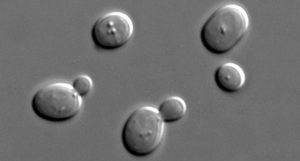
Uncovering your yeasts’ concentration and viability is important information for brewers, distillers and winemakers. The results can drastically influence what action to take during fermentation, and can have consequential impacts on the fermentation, flavor profile and the cost of ingredients for your fermented beverage. However, the process to do so is difficult, tedious and timely. Further complicating the matter is the counting of budding cells which require even more expertise.
Not taking into consideration the number of budding cells is fraught with folly. Not only is the percentage of budding cells important to know, the final concentration can be inaccurate if budding cells are not considered.
So, what are budding cells, why are they important and how do you count them?
What are budding cells?
Saccharomyces Cerevisiae reproduce asexually where the nucleus of the mother cells will split into a daughter cell. The daughter cell will grow until it is big enough to separate from the mother. One cell has become two.
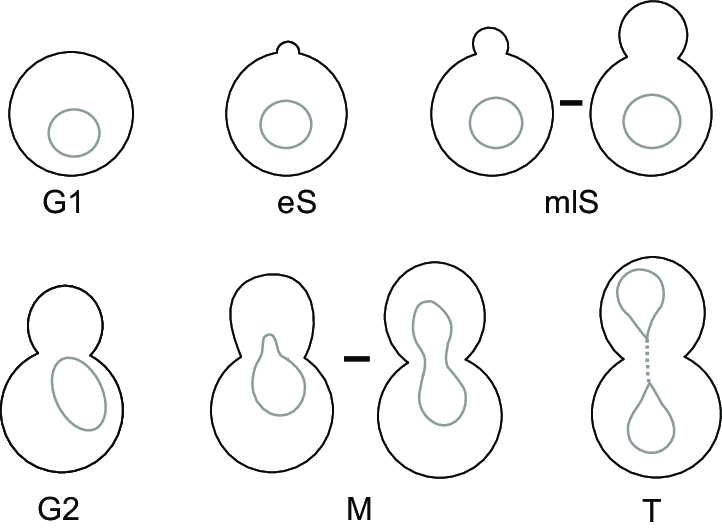
What happens during budding and how to count them?
The early stages of budding are very energy intensive, particularly for the mother cell. Think of it like raising children – when they’re young, they require a lot of energy and support, whereas as they get older, they require less support and eventually go out on their own.
We have to consider this when counting budding cells and decide at which point we should count the budding cells as one cell and when we should count budding cells as two cells. Remember, when the budding cells (children) are young, they are very fragile, so their likelihood for survival is lower.
There are several scientific opinions on this. The American Society of Brewing Chemists (ASBC) recognize a budding cells as two distinct cells once the daughter cell reaches 50% or more in the volumetric size of the mother cell 1. Any less than this, and the mother/daughter is counted as one cell. Another perspective is from the Australian Wine Research Institute (AWRI) which views the budding relationship as two cells once the daughter reaches 1/3 the volumetric size of the mother cell 2.
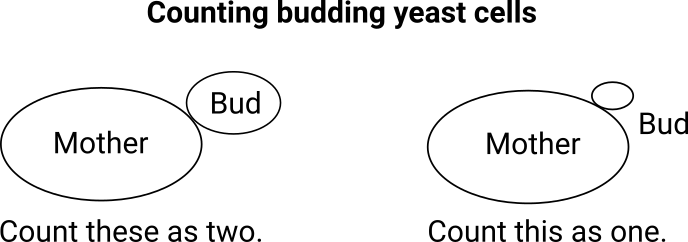
Instructions on counting yeast from the Australian Wine Research Institute 2
This can be complicated when counting yeast manually. Regardless of which method you are using, you first need to identify whether the cells are budding, and then judge whether to count them as one cell or two. Effectively, this assessment can be quite subjective as you need to eyeball the size and make a judgment call. Judging by height would be hard enough, but in this case, you are making a judgement call on the volume! This is the beauty of automated counting that can count budding cells – not only are the budding cells more easily identified, they are accurately measured.
How does staining affect budding cells?
Staining is done to test whether yeast are viable or not (living or dead). A toxic staining agent, frequently methylene violet or methylene blue, is mixed with a yeast sample where it is quickly absorbed. Living cells will force the staining agent out of the cell while the dead cells will not, and as such, will appear stained under a microscope 3. The incubation period for this reaction is five minutes. As the staining agents are toxic, eventually the yeast will succumb and absorb the staining agent, signaling death.
Budding cells are not able to excrete staining agents and as such, many appear stained under a microscope. However, as budding cells, they are actually living so the stain is actually deceiving. Therefore, these cells must be counted as being viable. This further complicates the manual counting process.
What we can learn
We spoke to Dr Tony Balzan (personal communication, December 5, 2019) from yeast producer AB Biotek about what we can learn from budding cell counts.
“The number of budding yeast cells can tell us a lot of what is happening during fermentation. Firstly, during the early stages of fermentation, a high budding percentage is indicative of good start yeast vitality and a healthy fermentation. Early on, we are expecting our cell sizes to be smaller as young daughter cells quickly initiate new cycles of replication. As fermentation progresses and environmental stresses increase (e.g. ethanol concentration and low pH) yeast energetic status deteriorates and the budding percentage will drop. By the end of fermentation, single cells become noticeably larger as yeast move into survival mode, while the budding cell percentage becomes very low.”4
A high budding cell percentage is also very important during the tirage process or for winemakers who are building up their yeast for pitching. A low budding percentage during these processes will inevitably lead to problems later on.
When repitching yeast, look for larger, single cells with a high viability percentage well above 90%.
Review
Shockingly, most automated cell counters cannot recognize budding cells. As a result, their viability and concentration results can be wildly inaccurate which does not help the winemaker/brewer/distiller. To get the most accurate results possible, an automated cell counting system must be able to identify and consider budding yeast cells. Oculyze BB2.0 and Oculyze FW are both automated yeast cell counting systems which specialize in the counting of budding yeast cells.


For more information, drop us a line at [email protected]
Also, you can now get access to a fully functional demo account to test our Web App. Completely free of charge and with no commitment to purchase.
References
1 – American Society of Brewing Chemists. Microscopic Yeast Cell Counting. Available: https://www.asbcnet.org/lab/getstarted/Documents/Yeast-4.pdf>>
2 – The Australian Wine Research Institute (2020). Total & Non-viable Counts of Yeast Cells in a Culture. Available: https://www.awri.com.au/industry_support/winemaking_resources/laboratory_methods/microbiological/identification-of-yeast/non-viable_counts/> 2019.
3 – Kwolek-Mirek, M., & Zadrag-Tecza, R. (2014) Comparison of methods used for assessing the viability and vitality of yeast cells. FemS Yeast Research. 2014;14(7): 1068-1079. doi.org/10.1111/1567-1364.12202
4 – Personal conversation with Dr Tony Balzan of AB Biotek, Australia.
Stay on top on important fermentation insights – subscribe to our monthly newsletter and receive a hand-picked selection of our most relevant articles straight to your inbox.
Never miss a beat and get real time updates with a new article each workday by subscribing our social media channels.
Instagram | Facebook | Twitter | YouTube


